An acid-base titration is a significant chapter for chemistry. The article is going to discuss this matter in a brief format.
Disclaimer: We must be clear that it is the subject of Chemistry. For this reason, we need to use the sign language of pure character. We are also taking help from a trusted site and online books. For this reason, you can find many similar equations, terms and processes.
What is Titrations
Titrations can identify hundreds of chemical and inorganic substances based on how acidic or basic they are. A base and an acid are titrated one with the other. Adding an indication typically allows for endpoint detection.
Theory of weak acid, strong base titration
Strong or weak acids or bases are involved in acid-base titration. An acid-base titration can be used to determine the following in particular.
The quantity of either an acid or a base
The strength or weakness of an unidentified acid or base.
pKa of an unidentified acid or pKb of an unknown base
Take an acid-base reaction with a proton acceptor as an example.
The proton typically dissolves as H3O+ in water.
Water is added to the base to acquire (H3O+) or lose (OH-).
Reversible acid-base reactions exist.
The solutions are given below.
H3O+ + A- = HA + H2O (acid)
H2O + B- = BH + OH- (base)
Here, the conjugate base is [A-], and the conjugate acid is [B-H].
Conjugate base + Conjugate acid = Acid + Base
Critical factors of weak acid strong base titration
Titration is adding a known-potency solution to a pre-treated sample containing an indicator in a predetermined volume.
Titrant: A solution with known concentration and strength used in the titration.
Titrand – Any solution that has the titrant added to it and contains the ion or molecule being measured is referred to as the titrand.
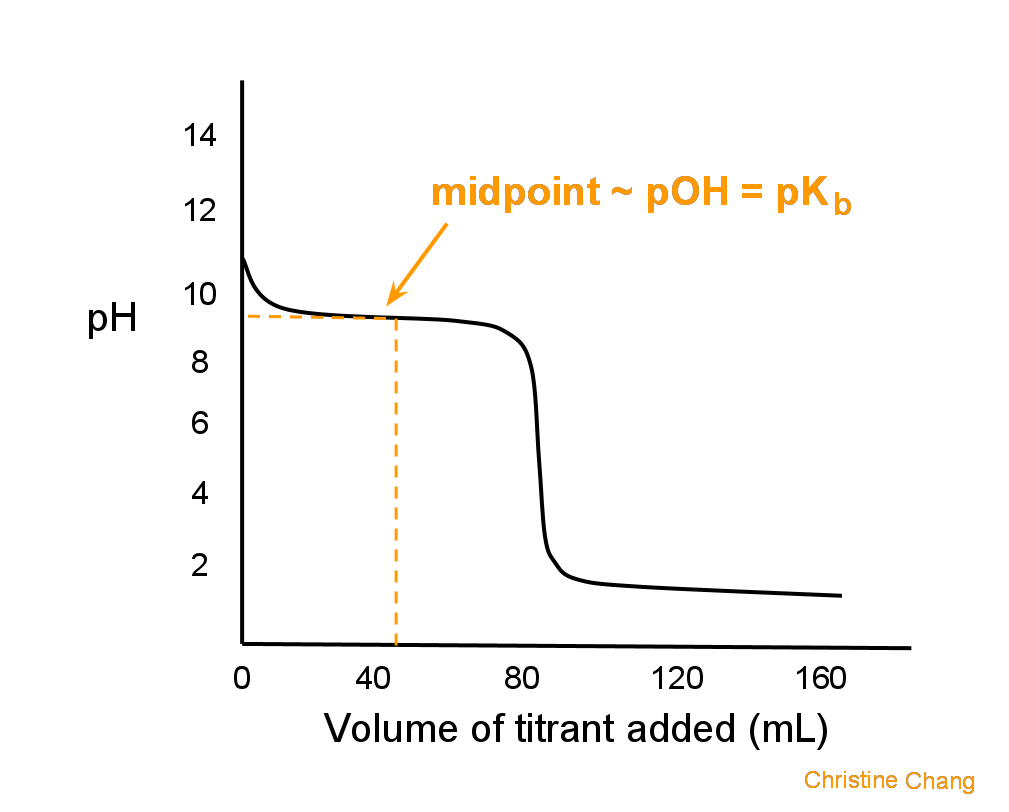
A titration curve is a plot of pH vs titrant volume that demonstrates how titrant volume affects pH during an acid-base titration.
The point at which just enough reagent is added to completely react with the material is known as the equivalence point.
A buffer solution resists pH alterations despite adding a potent acid or base or diluting it with water.
Know about acid base titration
Strong base/strong acid sodium hydroxide with hydrochloric acid
Diluted acid, robust base
Acid ethanol and sodium hydroxide
Strong base-weak acid
Both ammonia and hydrochloric acid
Weak base-weak acid Ammonia and ethanol
The indicators of weak acid strong base titration
Acid-base indicators are compounds that, at a specific pH, change colour or become turbid. They measure pH in addition to locating the equivalency point. They exhibit significant colour changes, are soluble, and are themselves acids or bases.
They have organic makeup.
An electron isomerism resonance causes the colour change. Varying indicators exhibit a change in colour at various pH intervals because they have different ionisation constants.
Three general categories can be used to organise acid-base indicators.
Sulphophthaleins and phthaleins (eg; Phenolphthalein)
Indicator azo (eg; Methyl orange)
Indicator phenylmethane (e.g., Malachite green)
The indicator of weak acid strong base titration
Strong base/strong acid – Due to its easier-to-detect colour change, phenolphthalein is typically selected.
Weak base, weak acid Both methyl orange and phenolphthalein are inappropriate.
Since an indicator needs a vertical section of the curve spanning two pH units, they need to be more adequate.
Phenolphthalein is employed and changes dramatically at the equivalence point, making it an excellent choice for weak acid/strong base reactions.
Methyl orange will vary significantly at the equivalency between a strong acid and a weak base.
Other Facts
- Titrations occur when two properly selected reactants, in this case, a strong base and a weak acid, come together. A titration curve displays the pH change during titration and indicates the potency of the associated acid and base. The titration curve showing how the pH changes when a strong base is added to a weak acid demonstrates that the pH changes initially quite slowly and gradually. This indicates that a buffer system forms as the titration gets closer to the equivalence point.
- At the start of the titration, the pH of the solution is nearly equal to that of the weak acid in water. The weak acid is wholly neutralised and changed into its conjugate base at the equivalence point (the number of moles of H+ = the additional number of OH-). The pH at the equivalence point, though, is not 7. This results from the conjugate base being produced during the titration. The solution that emerges is somewhat simplistic. The reaction’s stoichiometry determines the equivalence point, whereas the endpoint is the indicator’s colour change. The two issues are different.
- Stoichiometry Calculating and studying the quantitative (measurable) correlations between the reactants and products in chemical reactions (chemical equations).
- Buffer is a substance that balances a liquid’s acidity (pH).
- pH is a measure of a substance’s acidity or alkalinity that ranges in numerical values from 0 (highest edge) through 7 (neutral) to 14; the negative of the logarithm to base 10 of the concentration of hydrogen ions, measured in moles per litre (maximum alkalinity).
- Comparison point to the stage of a chemical reaction where an equal amount of acid and base have been combined.
The limitations of weak acid strong base titration
You want to achieve specific values when titrating a weak acid and a strong base. The initial pH, the pH following a small amount of base addition, the pH at half-neutralization, the pH at the equivalence point, and the pH following excessive base addition are among them.
This information will be adequate to understand the titration.
Here is an illustration of this procedure.
In the titration of 25 mL of 0.3 M HF with 0.3 M NaOH, determine the pH at each of the following steps.
The value of ka is 6.6 104.
The starting pH
0.3 M NaOH in 10 mL is then added.
0.3 M NaOH in 12.50 mL is then added.
0.3 M NaOH in 25 mL is then added.
0.3 M NaOH in 26 mL is then added.
We have discussed all the basic facts of a weak acid and strong titration. As we told you, it is the fundamental thought of Chemistry. So understanding the equation is a must.
Read more blog: Learn how much of the ocean has been explored